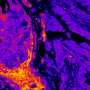
A groundbreaking study published in Nature Communications reveals that cancer cells possess a distinct metabolic fingerprint linked to their nuclear DNA. Researchers found that over 200 metabolic enzymes, typically involved in energy production within mitochondria, also localize directly on human DNA. This discovery suggests that various cell types, tissues, and even different cancers exhibit unique patterns of these enzymes, marking the first evidence of what the authors define as a “nuclear metabolic fingerprint.”
The research team conducted extensive analysis to demonstrate how specific metabolic enzymes are compartmentalized within the nucleus of cells. This positioning allows these enzymes to interact directly with DNA, potentially influencing genetic expression and cellular behavior in ways previously unrecognized. The implications of this finding could be significant for understanding cancer biology and developing targeted therapies.
Understanding the metabolic landscape of cancer cells is essential for identifying how they differ from normal cells. The study emphasizes that the metabolic profiles are not uniform but vary greatly among different types of cancers and tissues. This variability suggests that personalized treatment strategies may be necessary, as therapies could be tailored based on the unique metabolic fingerprints of a patient’s cancer.
The research adds to a growing body of evidence highlighting the importance of metabolism in cancer progression. By illuminating the intricate relationship between metabolism and genetic material, scientists hope to pave the way for innovative diagnostic tools and therapeutic approaches.
As cancer treatment evolves, understanding these metabolic fingerprints could also lead to the identification of new biomarkers for early detection. This could enhance the effectiveness of existing treatments and potentially improve patient outcomes.
In conclusion, the discovery of a nuclear metabolic fingerprint in cancer cells opens new avenues for research and treatment. As further studies build on these findings, the potential to redefine cancer therapy approaches becomes increasingly tangible. The work underscores the critical need for ongoing research in metabolic pathways and their roles in cancer dynamics.